Combustion Reaction ( Hydrogen) For this experiment our class was going to learn how elements work and how to get hydrogen by itself. The scientific Method Question: Will the balloon rise up to the ceiling? Materials: Balloon, Magnesium, Citric acid, water, and beaker Experiment steps: 1. Andrew put citric acid into the beaker 2. After andrew told us to curl the magnesium strip 3. Drop the magnesium into the beaker 4. Then put the balloon on the top of the beaker so it can capture the hydrogen 5. Once the bottle isn’t hot tie up the balloon Experience: In the beginning of the experiment andrew gave us a background of elements, atoms, and Thermodynamics of chemicals. Andrew put citric acid in the beaker and then griffin roll up the magnesium so it can fit into the beaker. After he was finished roll them up we place them in the beaker. As soon as we put all the roll in we quickly place the balloon on top and watched it inflate. A while later I notice the beaker was heating up because of the reaction between the acid and the magnesium. After the beaker started cooling down that's how you know no more hydrogen inflate the balloon. The hydrogen smelled horrible. After that we tied it up played around with it. I let the balloon go and it reached neutral buoyancy. As Bryan delgado walked in he was amazed as he saw a balloon floating in place. Science Behind It: Atoms will act in such a way just to fill up there outer shell. The noble gases are inert meaning they will not interact with other element because it has no need for more electrons in their shells. All atoms want 8 electron to have a full shell. In the experiment the chemical reaction between the citric acid and the magnesium is interesting because it separates the hydrogen into the balloon. For this section our team created rockets. In this project my partner was griffin because we both had integrated a different project into chemistry. We decided to become partners since we were going to be busy with the soap project.
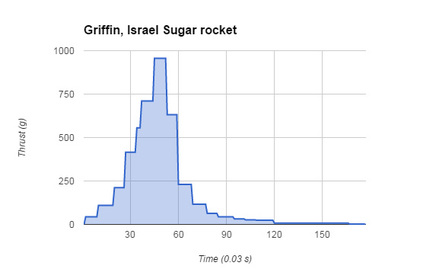
After we finished the soap project Griffin and I researched about rocket motors and fuel types. During the soap project we saw a video by Grant Thompson of his homemade sugar rocket which is made of potassium nitrate. We choose to do the sugar rocket because it burners a lot slower and because there wasn’t any more black powder. After we saw the video we research the basics about rocket motors and the types. Like whats a core burner? Or what's the difference between a core and an end burner? During the rocket project andrew came to class on the weekend to manufacture all the rockets. At this time Griffin and I haven’t made a motor design because we were so busy with the soap project that we were behind. We were creating a design that was similar to Noah's and Skye’s motor, so they let us have their data. The motor design is below. At the launch we stood 100 feet away from the motor just in case it cato. Noah and Sky named the motor sugar daddy because it was a potassium nitrate motor. Sugar daddy was expected to slowly burner. At the motor test most of the motors Cato. When we were heading back to class that day water just started pouring, good thing it rained at the end of the launch. After we got the data we used it to create a thrust curve. We tried downloading the video, but it said it was to big of a file. Andrew shorten all the video so we can download them. After he shorten the video I download it and imported it into adobe premier. This application slowed down the video to .30 of a sec. We recorded the data on google spreadsheets and made a thrust curve out of that. In our thrust curve we can see that it slowly starts to take off because it is a sugar rocket. At .45 seconds the motor reaches a thrust of about 950 grams which is the peak and then drops really quickly. The burnout time for the motor is 5.9 in 30 frames for sec and the total thrust in grams is 30336. Scents
For this experiment we are extracting essential oil from juniper, to add to our soap., The scientific Method Question: Will we extract lots of juniper oil? Materials: Vile, Juniper, Dry ice, Pressure tank, Blow dryer, Scissors, and Funnell Experiment steps: 1. Gather all the materials 2. Cut juniper into tiny pieces and place in container 3. input the juniper into the pressure tank 4. Add dry ice until full 5. Ciel the pressure tank 6. Soon the tank will frost from the outside, so go to the sink and heat it up with hot water 7. Once the the tank has reached a PSI of 1,200 take it to the table for the next step 8. Setup the apparatus and place the Pressure tank upside down 9. Under the tank place a vial where the juniper oil will fall into 10. Then release the pressure slowly and you will see dry ice falling into the vial 11. Blow dry the vial to separate it from the essential oil Experience: In the beginning of the experiment I thought we were going to receive about 2-3 drops of oil, but we actually extracted about 5-6 drops. The stronger the scent the more oil it will extract. This experiment was very easy because we have done this exact experiment before. We hit some bumps when doing this extracting like lack of materials and other experiment to deal with. When I open the container the smell of juniper immediately hit me. Last time we did this experiment we were watch more than doing. This time Griffin and I were able to release the pressure and blow dry the veil, which was more engaging than watching. When doing this myself I understand now what andrew meant when he said “We're losing it”. I thought he meant we were losing the pressure instead of the oil. After the experiment the veil contained juniper which was yellow. Science: The science of this experiments starts when we seal shut the pressure tank. When we seal shut the pressure tank I realized it's no different from when andrew told us not to cover the top of the beaker. The reason why the tank doesn’t exploded is because it’s created to hold all the pressure that is building up. Once there is enough pressure for the dry ice to turn into liquid CO2 it will dissolve the juniper and extract all the oil. Soap and Cast
For this experiment we integrated Humanities and Chemistry. In this experiment we will create soap and shape it into a hand, for humanities purposes. The scientific Method Question: Will the soap be a success even though we changed the directions? Materials: 5 Oz Olive oil, 10 Oz Coconut oil, 4.5 Oz Water, 2.39 Oz Pure Lye, Essential oil juniper, Goggles, Metal pot, Experiment steps: 1. Measure all the ingredients 2. Place coconut oil on slow cooker 3. When coconut oil is finished melting, add the olive oil 4. Then separately add the lye to water 5. Add the lye/Water mixture to the pot 6. Let it cook on high for 3 minutes 7. After pour the substance in a blender 8. Blend until you see a trace 9. Then pour the substance into the “cast” (Glove) Experience: We have been waiting to do this experiment for quite some time. Honestly this experiment was boring and to complicated. The reason why this was boring was because it went so fast and we had to think about the next step. As we setup the materials we had technical difficulty. Our computer was dead and there wasn’t any others left to use. My group was forced to charge the computer far from the experiment table making it harder to get the next step quicker. I never realized how complicated making soap was. What struck me the most was how long it takes for soap to cure (2 weeks). When the soap was ready to mold andrew decided take one for the team and help fit the soap into the glove. He was exposed to the lye in the soap and said “ It feels like burning cuts. After he fit the soap in the cast he walk in rush towards the sink to get rid of the burning pain. Science Behind It: When my partner and I decided to make soap we didn’t expect there to be a lot of science to it. Soap is a water soluble thats is made of oils. Soap molecules have two parts, ionic heads and hydrocarbon head. These two molecules are like ying and yang. Ionic head formed from metal components of hydroxide. This part of the molecule is hydrophilic meaning it will interact with water. Hydrocarbon tail is the opposite of the ionic head. Hydrocarbon tail does not like interacting with water. What happens when soaps are put to action is that the ionic molecule helps absorb all the unwanted fat and oils and while this is happening the hydrocarbon tail is as far from water. The hydrocarbon turns all thoses unwanted fats and oils into bubbles making it easy to get rid of. For a motor there is two types, a core burner and an end burner. A core burner has a hollow core and if the core’s length is two short then it may not be successful and if it is too long then the chambers pressure may exceed and cato. A end burner is easier to manufacture due to the way it is designed. Ender Burners have a faster burn time and are a full packed motor. This type of motor produces a constant level of thrust. The down side to end burners is that they have a chance exploding because the casing inside is unprotected from the flame. There are two types of motor fuels that we had the option to choose: Potassium nitrate and Black powder. Black powder burns fast and can exert a lot of thrust. Black powder can help reach a high altitude really quickly. A potassium nitrate motor or sugar motor burns slower in comparsion to a black powder motor.
|
|